The Fluent® system is designed to transform the fluid management experience through a streamlined workflow in the operating room.

No More Tangling With Tubes
The Fluent system was designed using input from healthcare professionals who have endured the complexities of fluid management firsthand. Its intuitive operation can streamline hysteroscopic procedures for the entire OR team.
Simplified setup and operation
- FloPak™ cartridges snap into place for simple setup.
- Intuitive touchscreen automatically guides setup and operation.
- Single-waste bag design eliminates the need for multiple canisters.
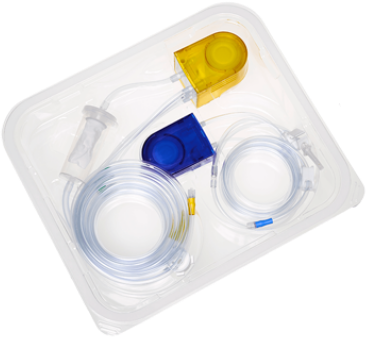

Advanced technology designed to increase clinical confidence
- Accurate fluid deficit readings within +/- 50 mL (1.69 oz)¹
- Advanced pressure Control maintains consistent intrauterine distention²*
- FloPak technology helps manage fluid use throughout the procedure³
*When set to 120 mmHg.

Streamlined workflow for the OR team
- Visual and audible alerts keep the procedure moving efficiently.
- Procedure kits give nurses all the components they need in one SKU.
- Less equipment required eliminates need for separate controls for the Fluent and MyoSure® systems.

Hear From a Fluent User
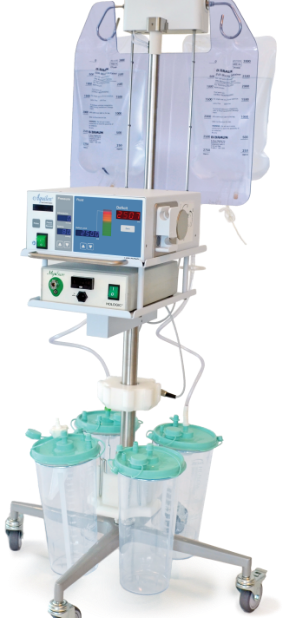
The Aquilex® Fluid Management System
Optimized for Control
The Aquilex system is designed to optimize high-aspiration procedures such as MyoSure tissue removal, and help you maintain control.
Feel Confident With Aquilex Technology

The Aquilex fluid management system includes:
- A non-weight-based inflow volume measurement system that reduces the risk of inaccurate deficit readings
- The ability to add or remove bags without pausing the system
- A system pause function that stops both inflow and suction pumps and locks the fluid deficit reading when a canister change is necessary

Unique features of the Aquilex system
- One integrated system with a fluid pump capable of 800 mL/min inflow
-
Two integrated vacuum pumps:
• One for the under-buttock drape and scope outflow capable of producing up to 125 mmHg of suction
• One for the MyoSure system capable of producing up to 500 mmHg of suction
- Automatic lumen calibration to optimize intrauterine pressure for achieving and maintaining distention and visualization
- Dual-scale technology means there is a scale for the supply hooks and one for the canisters for accurate deficit measurements
Hear OR Nurses Talk About the Importance of Fluid Management
Feature
Visit Our Virtual Surgery Suite
See how our products work in a three-dimensional surgery suite.
Important Safety Information
The MyoSure® tissue removal system, consisting of the MyoSure tissue removal devices (LITE, REACH, XL) and MyoSure controller, is intended for hysteroscopic intrauterine procedures by trained gynecologists to resect and remove tissue including submucous myomas, endometrial polyps, and retained products of conception. The MyoSure MANUAL hysteroscopic tissue removal device is intended for intrauterine use by a trained gynecologist to hysteroscopically resect and remove tissue, including focal lesions such as endometrial polyps and retained products of conception. MyoSure products are not appropriate for patients who are or may be pregnant, or are exhibiting pelvic infection, cervical malignancies, or previously diagnosed uterine cancer.
For more details on risks and benefits of the MyoSure system, MyoSure MANUAL tissue removal device, MyoSure hysteroscope and the Aquilex® fluid control system, please consult their respective IFUs.
The Fluent® fluid management system is intended to provide liquid distension of the uterus during diagnostic and operative hysteroscopy, and to monitor the volume differential between the irrigation fluid flowing into and out of the uterus while providing drive, control and suction for hysteroscopic morcellators. The Fluent fluid management system may not be used to introduce fluids into the uterus when hysteroscopy is contraindicated. The system should not be used to remove pathologies from pregnant patients or patients exhibiting pelvic infection, cervical malignancies, or previously diagnosed endometrial cancer. For detailed benefit and risk information, including contraindications relative to endometrial ablation, please consult the Instructions For Use.
1. Hologic, Inc. Data on File, bench testing. VER-07696, per 3000mL bag.
2. Hologic, Inc. Data on File, bench testing. VER-08393.
3. Hologic, Inc Data on File, VER-08393, Sample size: N=20 with MyoSure REACH TRD running, does not include priming or procedure time when MyoSure TRD is not running). Does not include priming or procedure time when MyoSure is not running.