NovaSure® endometrial ablation is the one and only gold standard in ablation technology.



Abnormal uterine bleeding (AUB) can dramatically affect a woman’s comfort, confidence, and quality of life. The NovaSure® endometrial ablation procedure is a one-time, 5-minute procedure to reduce or stop menstrual bleeding for women with AUB.
Introducing NovaSure V5 Endometrial Ablation
Simple.
NovaSure V5 features a simple device replacement guarantee.
Accurate.
The addition of sheath markings designed to increase the accuracy and confidence of device seating and placement.
Customized.
SureClear™ technology provides constant contact with the array while suctioning vapor, blood and desiccated endometrium.
SmartDepth™ technology monitors and measures tissue impedance for a customized depth of ablation for each patient.
Smarter. Smaller. Customized.
Smarter.
The only ablation modality featuring SmartDepth™ technology and impedance-based GEA with a physiological endpoint. It continuously monitors and measures tissue impedance to ensure a customized and reproducible depth of ablation.
Smaller.
The NovaSure ADVANCED device requires 14% less dilation¹ than a competitive GEA device and features rounded tips in an innovative 6-mm sheath for ease of insertion.2
Customized.
NovaSure endometrial ablation features a unique Moisture Transport® system powered by integrated suction, providing constant tissue contact with the array, even in irregular anatomy.
The NovaSure endometrial ablation procedure is a simple way to reduce or stop AUB for improved comfort, confidence, and quality of life.
5 Minutes. 1 Procedure.
The NovaSure procedure is a quick, one-time procedure with an average treatment time of 5 minutes that can performed at any point in your patient’s cycle without any pretreatment required.
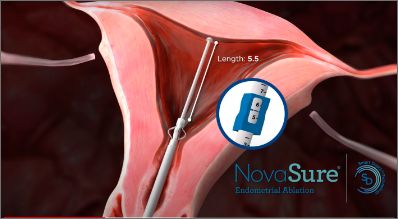
The SureSound® Uterine Cavity Measuring Device allows you to obtain cavity length with one measurement.
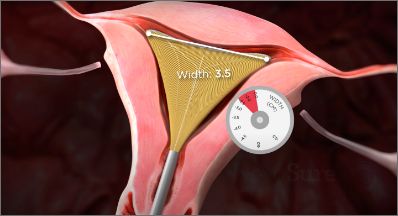
The NovaSure device includes a width dial for uterine width measurement and a customized ablation.
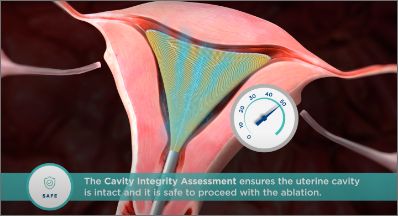
The Cavity Integrity Assessment is a proactive safety measure that ensures the uterine cavity is intact and it is safe to proceed with the ablation.
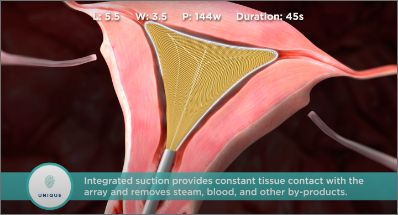
The unique Moisture Transport™ fluid removal system with integrated suction provides constant tissue contact with the array and removes steam, blood, and other by-products even in cases with irregular anatomy.
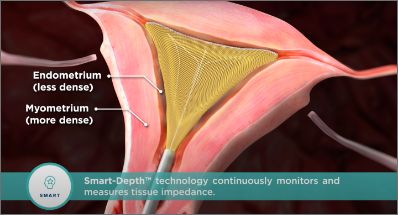
With SmartDepth™ Technology, as RF energy ablates the endometrial lining and approaches the myometrium, it will reach a higher level of impedance that it can no longer penetrate. The density of the muscle provides a physiological endpoint allowing the NovaSure controller to sense the resistance and stop the energy, completing the ablation.
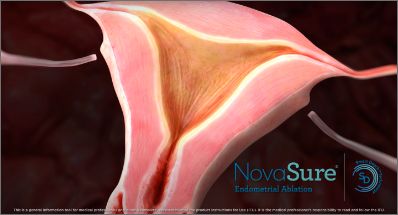
The completed procedure shows a contoured depth of ablation specific to each patient’s unique anatomy.
NovaSure Testimonial with Dr. Bethany Peterson
20 Years of Innovation
Feature
Visit Our Virtual Surgery Suite
See how our products work in a three-dimensional surgery suite.
Important Safety Information
NovaSure® endometrial ablation is for premenopausal women with heavy periods due to benign causes who are finished childbearing. Pregnancy following the NovaSure procedure can be dangerous. The NovaSure procedure is not for those who have or suspect uterine cancer; have an active genital, urinary or pelvic infection; or an IUD. NovaSure endometrial ablation is not a sterilization procedure. Rare but serious risks include, but are not limited to, thermal injury, perforation and infection. Temporary side effects may include cramping, nausea, vomiting, discharge and spotting. Inform patients to contact you if they experience a possible side effect related to use of this product. For detailed benefit and risk information, please consult the IFU.
- NovaSure ADVANCED Instructions for Use, 6mm dilation as compared to Minerva Instructions for Use 7mm dilation
- Based on internal bench testing that measured perceived ease of insertion through a simulated cervix of both the 6 mm and 8 mm devices. N=16 surgeon users.
- NovaSure ADVANCED Instructions for Use.
- Hologic, Inc. Data on file; 2004-2018. Based on units shipped from 2004-2018.

