Explore the virtual MyoSure® Learning Lab to understand pathologists’ perspectives on sample quality when utilizing a MyoSure tissue removal device.

Help women with abnormal uterine bleeding avoid the trauma of major surgery with the MyoSure® tissue removal procedure, a hysteroscopic treatment that removes uterine tissue, including fibroids and polyps.
Choose the Right Device for the Right Procedure
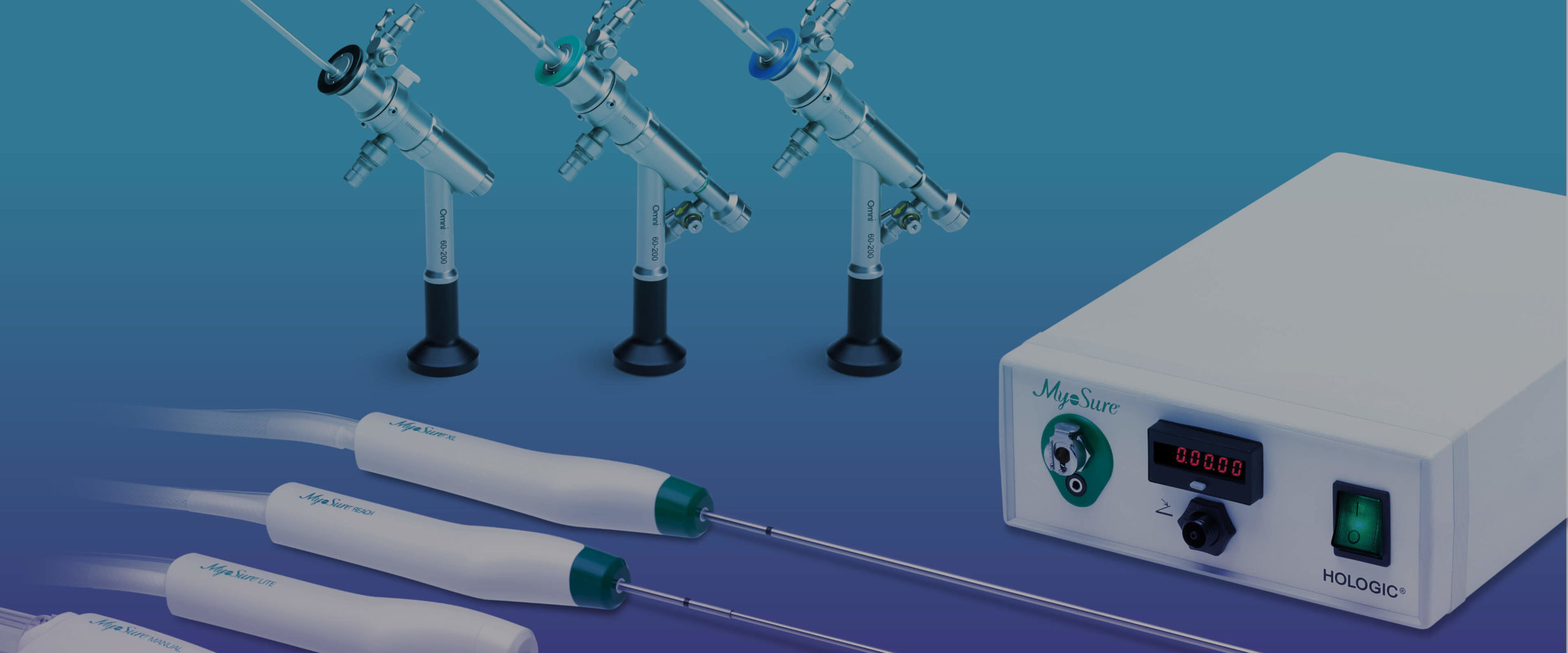
The full suite of MyoSure devices gives you a solution for tissue removal in a wide range of sizes and locations. For women who would like to have children in the future, the MyoSure procedure may be a treatment option to consider.

MyoSure XL device: Designed to optimize cutting efficiency with a large cutting window and suction on demand.
MyoSure REACH device: Access hard-to-reach pathology to help improve operative efficiency.
MyoSure LITE device: Visualize the entire uterine cavity to get a quality tissue specimen; remove small polyps up to 3 cm with minimal dilation.
MyoSure MANUAL device: Perform in-office polypectomy and targeted biopsy under continuous visualization with the Hologic hysteroscope without the complexity, logistics, and costs of the operating room.
Preserve What Matters Most: The Uterus
The MyoSure procedure is simple and efficient, and it can be performed in-office.
Clear visualization: High-quality optics afford a direct view of the uterine cavity, so you can easily see the anatomy and guide the procedure from start to finish.
Resect uterine tissue: While the stainless-steel blade resects the unwanted tissue, the fluid management system simultaneously removes it from the uterine cavity using suction, providing a clear field of view.
Side-facing cutting window: Optimized to provide better tissue contact for efficient removal and to minimize risk of perforation.
Preserves uterine function: Resection is done mechanically. Because no RF energy is used, uterine form and function are preserved and collected specimens remain intact.
See MyoSure in Action
Educating Your Patients
In addition to bringing you best-in-class technology and support, Hologic has developed an online portal for Patient Marketing Materials, providing you with ready-to-print and downloadable materials to help streamline your patient outreach and education initiatives.
Feature
Visit Our Virtual Surgery Suite
See how our products work in a three-dimensional surgery suite.
Important Safety Information
The MyoSure® hysteroscopic tissue removal system is intended for hysteroscopic intrauterine procedures by trained gynecologists to resect and remove tissue including submucous myomas, endometrial polyps, and retained products of conception. It is not appropriate for patients who are or may be pregnant, or are exhibiting pelvic infection, cervical malignancies, or previously diagnosed uterine cancer.
